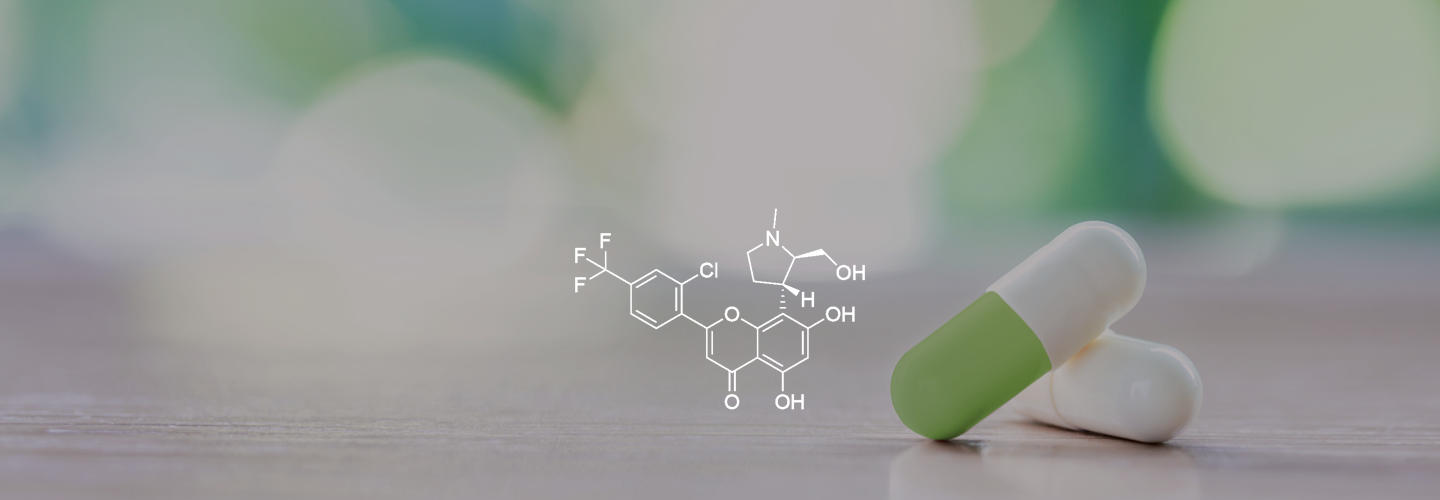
Cyclin-Dependent Kinase (CDK) 9 Inhibitor
Voruciclib is a selective orally administered CDK9 inhibitor with potential to treat both hematological malignancies and solid tumors.
Voruciclib, a selective orally administered, inhibitor of CDK9, is in clinical development for acute myeloid leukemia. Applications in solid tumors are also being evaluated in non-clinical models.
CDK9 has important functions in cell cycle regulation, including the modulation of two therapeutic targets in cancer: myeloid leukemia cell differentiation protein ("MCL1") and the MYC proto-oncogene protein ("MYC") which regulates cell proliferation and growth. Voruciclib is currently being evaluated in a Phase 1 trial evaluating dose and schedule in patients with relapsed or refractory (R/R) acute myeloid leukemia ("AML"). Applications in solid tumors are also being considered where MYC is dysregulated.
Additional Information
The CDK family of proteins are important cell cycle regulators responsible for the control of cell proliferation, differentiation, apoptosis, and DNA repair. CDK9, one of several members of the CDK family of proteins, functions as a gene transcription controller and is also involved in regulating protein degradation. Specifically, CDK9 is a promising target to treat a range of cancers because of its role in controlling two other proteins often dysregulated in cancerous cells: myeloid leukemia cell differentiation protein (“Mcl-1”) and the MYC proto-oncogene protein ("Myc").
- CDK9 is a transcriptional regulator of MCL1, a member of the family of anti-apoptotic proteins which, when elevated, may prevent the cell from undergoing cell death. Inhibition of CDK9 blocks the production of Mcl-1, which is an established resistance mechanism to the B-cell lymphoma ("BCL2") inhibitor venetoclax (Venclexta®).
- CDK9 is a transcriptional regulator of the MYC proto-oncogene protein ("Myc") which regulates cell proliferation and growth. Upregulation of MYC is implicated in many human cancers and is frequently associated with poor prognosis and unfavorable patient survival. CDK9, in addition to being a transcription factor for MYC, also decreases phosphorylation of MYC protein that is implicated in stabilizing Myc in KRAS mutant cancers. Targeting MYC directly has historically been difficult, but CDK9 is a promising approach to target this oncogene.
Our development program for voruciclib is exploring applications that leverage the role of CDK9 in controlling two proteins often dysregulated in cancerous cells: MCL1 and MYC.
Voruciclib: Inhibition of MCL1
In pre-clinical studies voruciclib shows dose-dependent suppression of MCL1; in December 2017 a study of voruciclib published in the journal Nature Scientific Reports reported that the combination of voruciclib plus the BCL-2 inhibitor venetoclax was capable of inhibiting two master regulators of cell survival, MCL-1 and BCL-2, and achieved synergistic antitumor effect in an aggressive subset of DLBCL pre-clinical models. Additional preclinical studies demonstrate that the inhibition of CDK9 by voruciclib synergistically enhances cell death induced by the BCL-2 selective inhibitor venetoclax in preclinical models of acute myeloid leukemia. (Sig Transduct Target Ther 5, 17 (2020). https://doi.org/10.1038/s41392-020-0112-3.
The research suggests voruciclib’s potential as an attractive therapeutic target for treating cancers in combination with venetoclax or other BCL-2 inhibitors, to address potential resistance associated with MCL1, and is supportive of our ongoing clinical program evaluating voruciclib. Currently, we are evaluating voruciclib plus venetoclax in patients with R/R AML in a Phase 1 clinical trial. The trial started with the evaluation of dose and schedule of voruciclib as a monotherapy in patients with R/R B-cell malignancies and AML after failure of prior standard therapies to determine the safety, preliminary efficacy and maximum tolerated dose. The trial is now evaluating the dose and schedule of voruciclib in combination with venetoclax, a BCL2 inhibitor, to assess synergies and the opportunity for combination treatments, initially in patients with R/R AML.
Initial results from the Phase 1 study have demonstrated anti-leukemic activity across multiple heavily pretreated patients along with anticipated decreases in Mcl-1. No evidence of overlapping toxicity, and no dose limiting toxicities have been observed to date. Favorable pharmacokinetics have been observed, including a half-life supporting once-a-day oral dosing, dose proportional C-max and a high volume of distribution suggesting broad entry into tissues.
Voruciclib: Inhibition of MYC
Dysregulation of MYC is associated with many cancers. CDK9 is a known regulator of MYC transcription and a modulator of MYC protein phosphorylation. Reported data in preclinical models demonstrates that voruciclib:
- Results in a rapid decrease in the phosphorylation of proteins that promote MYC transcription
- Rapidly decreases phosphorylation of MYC protein on Ser62, a site implicated in stabilizing MYC in KRAS mutant cancers
- Possesses single agent activity against multiple KRAS mutant cancer cell lines both in vitro and in vivo
- Synergistically inhibits KRAS G12C mutant cancer cell lines in combination with KRAS G12C inhibitors, both in vitro and in vivo
The research presented suggests that voruciclib could be an attractive therapeutic target for cancers driven by MYC overexpression such as those harboring KRAS mutations and warrants further investigation.
Clinical Program
In early clinical studies evaluating more than 70 patients with solid tumors in multiple Phase 1 studies, along with pre-clinical studies, suggests voruciclib’s ability to inhibit CDK9 at doses that appear generally well-tolerated to date in clinical studies. The ongoing development program is intended to further clarify the clinical dose and overall clinical profile of voruciclib, particularly for use in combination with other cancer therapies.
We are currently clinically evaluating voruciclib in combination with venetoclax in patients with R/R AML in a Phase 1 study. We have reported initial data demonstrating anti-leukemic activity, including complete responses in heavily pretreated patients. Additionally, at doses of 100 mg or more, initial results from correlative biomarker assay analyses of available samples from patients treated with the combination demonstrated anticipated decreases of myeloid leukemia cell differentiation protein (“Mcl-1”). Reductions in Mcl-1 are consistent with the known mechanism of action of CDK9 inhibition.
Initial data has also demonstrated that voruciclib in combination with standard dose venetoclax has been generally well tolerated with no dose limiting toxicities, and no evidence of overlapping toxicity, observed to date. The most common (≥5% of patients) grade 3 adverse events were myelosuppression associated with AML.
We are also continuing to evaluate the potential of voruciclib against cancers where MYC is dysregulated.
Voruciclib is being evaluated in patients with R/R AML. AML is a cancer that starts in the bone marrow, where the body produces new blood cells. It normally begins in cells that mature into while white blood cells, but can also start in cells that mature into red blood cells and platelets.
We are also considering the clinical evaluation of voruciclib in combination with other therapies in patients with solid tumors driven by MYC overexpression, such as those harboring KRAS mutations.