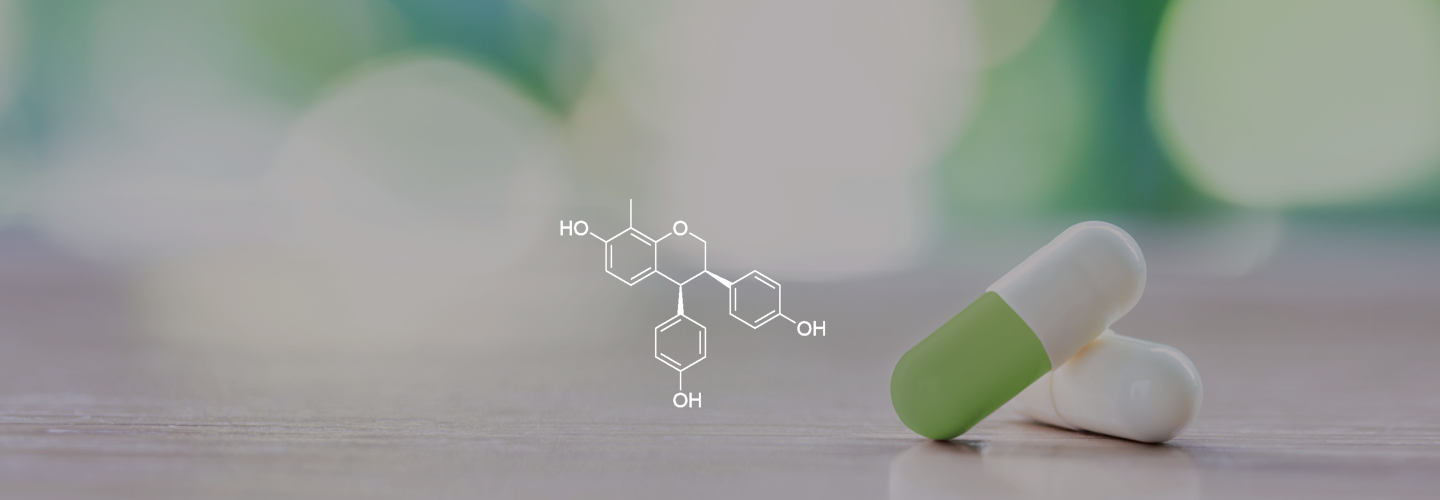
Inhibitor of Mitochondrial OXPHOS
ME-344, an inhibitor of mitochondrial OXPHOS, is a drug candidate with potential to treat solid tumors.
ME-344 is a novel drug candidate that in an inhibitor of mitochondrial oxidative phosphorylation (OXPHOS), a fundamental metabolic pathway involved in the production of adenosine triphosphate (ATP) in the mitochondria. Treatment of tumor cells with ME-344 as a single-agent results in a rapid loss of ATP and cancer cell death.
Although clinical investigation of ME-344 has demonstrated single agent activity in patients with solid tumors, using it in combination with other cancers therapies is thought to hold more significant potential for patients. Data reported from pre-clinical and clinical studies has demonstrated activity in combination anti-angiogenics, like the vascular endothelial growth factor (“VEGF”) inhibitor bevacizumab (AVASTIN®). ME-344 is continuing to advance via the ongoing development of a new formulation intended to improve biologic activity and patient convenience.
Additional Information
ME-344 is a novel drug candidate that is an inhibitor of mitochondrial oxidative phosphorylation (OXPHOS), a fundamental metabolic pathway involved in the production of adenosine triphosphate (ATP) in the mitochondria. ATP provides energy to drive many metabolic cell processes, including division, proliferation, and growth. By disrupting the production of ATP, ME-344 has been shown to induce cancer cell death in nonclinical models and was associated with antitumor activity in clinical studies.
The two main sources of ATP production in cells are OXPHOS and glycolysis; the latter is highly active in most tumors. Anti-angiogenics, like the vascular endothelial growth factor (VEGF) inhibitor bevacizumab (AVASTIN®), have the potential to normalize vasculature and decrease reliance on glycolysis. The resulting reduction in glycolysis may trigger an increased dependence on mitochondrial ATP production for energy to support continued tumor proliferation.
In such cases of tumor plasticity, the combination of ME-344 and bevacizumab may induce metabolic synthetic lethality, providing a novel therapeutic strategy. Specifically, leveraging the ability of antiangiogenics like bevacizumab to reduce glycolysis and force tumor cells to switch to mitochondrial respiration via OXPHOS, which is inhibited by ME-344, may reduce access to ATP needed for cell division and growth in tumors.
This approach was first clinically supported by a multicenter, investigator-initiated, randomized, open-label, window of opportunity clinical study, evaluating ME-344 (3 doses) plus bevacizumab (1 dose) in 42 women with early HER2-negative breast cancer. Study results demonstrated significant biological antitumor activity as measured by a reduction in the proliferative biomarker Ki-67 compared to placebo. The combination appeared to be generally well tolerated.
Further clinical support was provided by a Phase 1b study evaluating ME-344 in combination with bevacizumab in patients with relapsed metastatic colorectal cancer (“mCRC”) after failure of standard therapies. This study demonstrated clinical activity, including effect on progression free survival, in a cohort of 23 patients.
The data from this study were consistent with preclinical data suggesting that combining ME-344 can augment anti-angiogenic therapy and provided validation for continued evaluation of the combination of ME-344 with bevacizumab and other VEGF inhibitors.
An earlier Phase 1 clinical study evaluating ME-344 as a single-agent in patients with refractory solid tumors also demonstrated anti-tumor activity, further validating the potential of mitochondrial inhibition as a promising therapeutic modality.
ME-344 is continuing to advance via the development of a new formulation that is intended to improve biological activity and patient convenience of administration based on promising results from multiple clinical studies.
ME-344 clinical studies include a multicenter, randomized, open-label, clinical trial that evaluated the combination of ME-344 and bevacizumab an anti-angiogenic therapy, in 42 women with early HER2-negative breast cancer. Results the study were published in the November 2019 issue of Clinical Cancer Research.
The primary objective of the trial was to show proof of ME-344 biologic activity as measured by reductions in the nuclear protein Ki67 (expression of which is strongly associated with tumor cell proliferation and growth) compared to the control group who received bevacizumab plus placebo. Secondary objectives included determining whether ME-344 biologic activity correlates with vascular normalization. The data demonstrate significant biologic activity in the ME-344 treatment group:
- In ME-344 treated patients, mean absolute Ki67 decrease was 13.3 compared to an increase of 1.1 in the bevacizumab monotherapy group (P=0.01).
- In ME-344 treated patients, mean relative Ki67 decrease was 23% compared to an increase of 186% in the bevacizumab monotherapy group (P < 0.01).
Treatment was generally well tolerated; three grade 3 adverse events of high blood pressure were reported, two in the ME-344 arm and one in the bevacizumab monotherapy arm.
ME-344 was further evaluated in combination with the anti-angiogenic antibody bevacizumab (Avastin®) in a Phase 1b study evaluating patients with relapsed metastatic colorectal cancer (mCRC) with progressive disease after failure of standard therapies. Patients were administered ME-344 until disease progression or intolerance. The primary objective was progression free survival. Secondary endpoints included overall response rate, duration of response, overall survival, and safety.
Of the 20 evaluable patients enrolled in the study, 5 (25%) completed at least 16 weeks of therapy without disease progression, exceeding a predetermined objective of 4 (20%) patients as defined in the protocol. Nine (45%) of the 20 evaluable patients had stable disease. Further, the combination with bevacizumab at the dose and schedule evaluated was generally well tolerated with no overlapping toxicities observed.
Results from our earlier, first-in-human, single-agent Phase 1 clinical trial of ME-344 in patients with refractory solid tumors were published in the April 1, 2015 issue of Cancer. The results indicated that eight of 21 evaluable patients (38%) treated with ME-344 achieved stable disease or better, including five who experienced progression-free survival that was at least twice the duration of their last prior treatment before entry into the trial. In addition, one of these patients, a heavily pre-treated patient with small cell lung cancer, achieved a confirmed partial response and remained on study for two years. ME-344 was generally well tolerated at doses equal to or less than 10 mg/kg delivered on a weekly schedule for extended durations. Treatment-related adverse events included nausea, dizziness and fatigue. Dose-limiting toxicities were observed at both the 15 mg/kg and 20 mg/kg dose levels, consisting primarily of grade 3 peripheral neuropathy.
ME-344 has been evaluated in patients with solid tumors, which represent the most common type of cancer. Most solid tumors are carcinomas, which usually begin in the skin or the surface of internal organs and glands.
ME-344 is being evaluated in combination with bevacizumab, (marketed as Avastin®) in a Phase 1b study in patients with previously treated metastatic colorectal cancer.